Isocyanates are the unsung heroes of modern chemical engineering, playing a pivotal role in driving significant advancements across various industrial sectors. These highly reactive chemical compounds combine disparate elements to yield enhanced characteristics, including increased strength, exceptional resistance, and remarkable longevity in the materials they help create. Utilized in a broad range of applications from aerospace to automotive and heavy construction, isocyanates elevate the performance and efficiency of countless commercial products. The development of isocyanate-based materials involves a wide variety of processing techniques and chemical matrices, catering to both rigid structural requirements and flexible material properties. With capabilities that enable composites to be times stronger than conventional materials, isocyanates integrate into specific manufacturing processes to form durable sandwich structures and synthetic foams. The landscape of these essential chemicals, from their production costs and environmental sustainability to future market trends, is highly promising. Exploring how these revolutionary compounds will shape our industrial future becomes increasingly compelling for modern manufacturers.
What are Isocyanates?
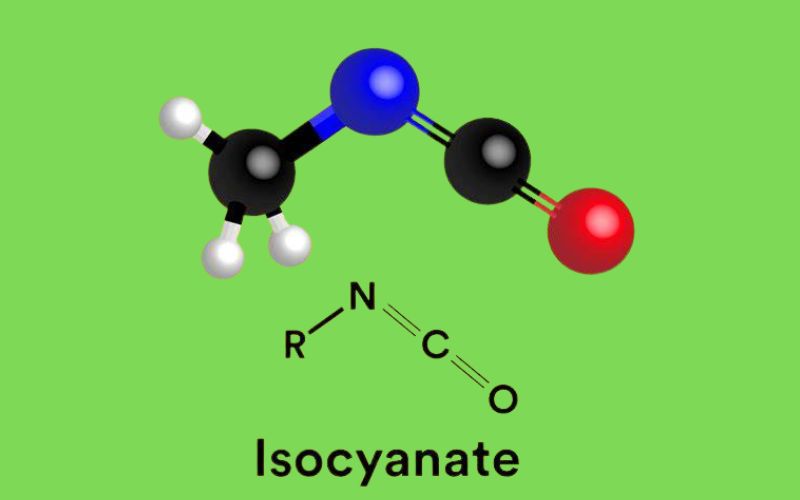
Isocyanates are engineered chemical compounds characterized by the presence of one or more isocyanate functional groups, typically represented by the formula -N=C=O. They serve as the foundational building blocks for a vast array of advanced materials, most notably polyurethanes. By reacting seamlessly with alcohols and other chemical constituents, they form complex polymer networks that can be several times stronger than individual natural materials. This unique chemical structure offers enhanced properties such as immense durability, structural flexibility, and high load-bearing capacity. The specific alignment and formulation of these molecules, analyzed through advanced chemical modeling, directly affect the resulting material’s behavior and physical properties, making them highly advantageous in various demanding industrial applications.
These versatile compounds can help create materials that exhibit either isotropic or anisotropic properties, depending on the orientation of the constituent polymer chains during curing. When engineers manipulate the isocyanate reaction, they can dictate whether the final product will stretch like rubber or stand firm like rigid plastic. They can easily incorporate continuous fibers, metal fillers, and other strengthening elements into the reaction mixture to achieve highly specific performance characteristics required by advanced manufacturing sectors.
The immense reactivity of the isocyanate group makes it a highly valuable tool in synthetic chemistry. Because they react readily with compounds containing active hydrogen atoms, isocyanates are incredibly versatile. This reactivity allows manufacturers to bond disparate materials together, creating resilient coatings, high-strength adhesives, and durable insulating foams. Ultimately, the unique chemical properties of isocyanates allow for the creation of customized, high-performance materials that drive innovation in modern product design and industrial engineering.
What Materials Constitute Isocyanates?
Isocyanates are composed of specific organic compounds that serve as the primary reactive agents in the formation of advanced polymers. These chemical structures typically consist of an organic radical attached to the functional -N=C=O group. Depending on the specific industrial requirement, these compounds are divided into distinct chemical families that offer varied performance benefits.
The primary building blocks include various types of monomeric isocyanates, such as aromatic and aliphatic compounds, which determine the fundamental strength and UV stability of the final polymer. Aromatic isocyanates, which contain a benzene ring, are primarily used for their rapid reactivity and high structural stiffness. They bind the reinforcement materials together quickly, making them ideal for heavy-duty foams and structural adhesives. Aliphatic isocyanates, on the other hand, lack the benzene ring and provide exceptional resistance to ultraviolet light, making them the preferred choice for exterior coatings and specialized outdoor applications.
To create functional materials, isocyanates are combined with matrix materials, such as complex polyols, to initiate polymerization. This essential chemical reaction forms the durable urethane linkages that give the resulting materials their signature strength. Additives, catalysts, and specialized chemical stabilizers are also introduced to control the reaction speed and enhance the physical properties of the final product.
Other key components include chemical blowing agents for foam production, flame retardants for safety compliance, and natural or synthetic fibers to reinforce the physical structure. The careful orientation of these fibers within the isocyanate-derived matrix creates isotropic and anisotropic behaviors, allowing engineers to produce materials that are up to 10 times stronger than their individual components. This precise combination of raw chemicals and targeted additives enables the creation of commercial isocyanate products specifically tailored for demanding modern industries.
How are Isocyanates Produced?
Isocyanates are produced through highly controlled chemical synthesis processes that require advanced industrial facilities to ensure safety and chemical purity. The most common commercial method involves the phosgenation of primary amines. In this complex process, a specific amine reacts with phosgene gas under strictly monitored temperatures and pressures to yield the desired isocyanate and hydrogen chloride gas as a byproduct. The raw chemical materials are prepared in various forms and continuously aligned to optimize the overall yield and structural purity of the final compound.
The selection and preparation of the precursor amines are critical for understanding the ultimate performance of the isocyanate. For instance, the production of Toluene Diisocyanate (TDI) requires nitrating toluene, followed by catalytic hydrogenation. The industrial production process involves advanced reaction vessels and separation columns, in which the upper and lower chemical phases are carefully separated to isolate pure isocyanate. Throughout this intense production cycle, mathematical relationships among chemical stress, thermal strain, and reaction forces are crucial for designing and optimizing the manufacturing infrastructure.
Because the chemicals used in this synthesis, particularly phosgene, are highly toxic, modern production facilities implement massive safety protocols. Closed-loop systems, continuous air monitoring, and automated emergency shutdown procedures ensure that the volatile compounds remain safely contained. Particle reinforcement and filtration systems capture any escaping emissions, protecting both the workers and the surrounding environment.
Once synthesized, the raw isocyanates undergo rigorous purification processes. Distillation and crystallization techniques remove any remaining chemical impurities, ensuring the final product meets exact industrial specifications. The pure isocyanates are then carefully stored in moisture-free environments, as they react violently with water, before being safely transported to manufacturers who transform them into the advanced composite materials we rely on every single day.
Types of Isocyanates
There are several key types of isocyanates that possess unique chemical properties and specific industrial applications. These distinct chemical families are carefully selected by engineers to ensure the final product meets exact structural and performance characteristics.
What are Aromatic Isocyanates?
Aromatic isocyanates represent the most widely used category of these reactive compounds in global manufacturing. The two primary examples are Methylene Diphenyl Diisocyanate (MDI) and Toluene Diisocyanate (TDI). These high-performance materials combine a rigid aromatic ring structure with high chemical reactivity, thereby significantly improving the mechanical properties of the resulting polymers. TDI is commonly used in the production of flexible foams for furniture and automotive seating, while MDI is widely used to produce rigid insulating foams and heavy-duty structural adhesives. The aromatic matrix binds the chemical chains tightly together, providing excellent shape retention and allowing the material to resist physical deformation and breakage under heavy structural loads.
What are Aliphatic Isocyanates?
Aliphatic isocyanates are engineered chemicals in which the isocyanate group is attached to an aliphatic carbon chain rather than an aromatic ring. The most common aliphatic variants include Hexamethylene Diisocyanate (HDI) and Isophorone Diisocyanate (IPDI). While they generally react more slowly than their aromatic counterparts, aliphatic isocyanates offer significantly superior properties in terms of light stability and weather resistance. These compounds do not yellow or degrade when exposed to direct ultraviolet sunlight. Consequently, aliphatic isocyanates are used extensively in high-end automotive clear coats, architectural paints, and industrial surface coatings, enhancing the performance in fields requiring long-term aesthetic durability and intense resistance to environmental wear and chemical corrosion.
What are Hybrid Isocyanates?
Hybrid isocyanates are advanced chemical engineering innovations that combine the rapid reactivity of aromatic compounds with the UV stability of aliphatic structures in a single application matrix, maximizing the benefits of each material. Examples include specialized prepolymers and blocked isocyanates used in advanced manufacturing. These hybrid composites can be used to create thermoplastic elastomers or highly cross-linked thermosetting plastics. Continuous chemical innovations employ targeted molecular adjustments to dramatically improve physical properties. Nanoscale chemical blending enables these hybrid isocyanates to be safely used in sensitive applications, offering high strength, low weight, and unparalleled chemical stability for the medical and aerospace industries.
Applications of Isocyanates in Modern Industries
Isocyanates, primarily as the core component in polyurethane production, find widespread and critical use in aerospace, automotive, marine, construction, sports equipment, renewable energy, and modern medical devices. Because they enable the creation of materials that are often stronger than traditional legacy materials, they offer immense advantages, including complete design flexibility, high resistance to structural corrosion, and highly tailored material properties. They can be used in combination with natural composites and organic matrices, making them highly versatile for a range of demanding applications.
Why are isocyanates used in construction?
Isocyanate-derived materials are widely used in modern construction for their high elastic modulus and exceptional insulating properties, which significantly enhance building strength and energy efficiency. Rigid polyurethane foams, made using MDI, are highly resistant to longitudinal fractures and can easily balance weak and strong structural phases to support heavy architectural loads. These composite insulation panels are built using various continuous lamination methods to achieve the desired structural stiffness, making them ideal for roofing, wall cladding, and commercial cold storage facilities. Key factors in their performance include the highly covalent nature of the isocyanate matrix, which enables them to withstand heavy physical loads while preventing heat transfer and moisture infiltration.
What benefits do isocyanates offer in aerospace?
In aerospace applications, isocyanate-based composites offer significant benefits by providing materials that are exceptionally lightweight yet times stronger than traditional aluminum structures. They consist of advanced matrix materials and individual synthetic fibers, offering advantages such as customized aerodynamic properties, high resistance to corrosion at high altitudes, and extensive interior design flexibility. The specific orientation of carbon fibers within an isocyanate-derived resin affects the isotropic or orthotropic behavior of the aircraft components. Factors like the quantity of material and the precise anisotropy heavily influence the safety and performance of the aircraft. These specialized composites are commonly used to manufacture interior cabin panels, overhead storage bins, and vibration-dampening engine mounts.
How do isocyanates enhance vehicles?
Isocyanates dramatically enhance modern vehicles by offering lightweight yet highly durable structural components that improve overall fuel efficiency, driving performance, and passenger safety. They are meticulously engineered with specific flexible and rigid forms to achieve desired properties such as high body stiffness and durable seating comfort. These advanced interior materials exhibit unique strain behaviors, including resistance to necking under crash impacts, which significantly contribute to the vehicle’s overall strength and reliability. The chemical reinforcement equations and stress-strain relationships play huge roles in designing automotive parts with optimal performance. From high-gloss exterior paints to acoustic dampening foams hidden within the dashboard, isocyanates create innovative solutions for global vehicle applications.
Physical Properties of Isocyanates
Isocyanates and the advanced composite materials they help produce exhibit a wide range of physical properties that make them highly versatile and desirable for various heavy-duty applications:
- Reactivity: Isocyanates are highly reactive compounds that readily form strong chemical bonds with alcohols, amines, and water. This intense reactivity is what makes them such effective building blocks for durable polymers.
- اللزوجة: Depending on the specific chemical structure, isocyanates range from low-viscosity, free-flowing liquids to high-viscosity, thick syrups. This adjustable property allows them to be easily sprayed, injected, or cast into complex molds.
- الاستقرار الحراري: Isocyanate-derived materials can exhibit excellent thermal insulation or high thermal conductivity, depending on the specific matrix used. This versatility allows for precise thermal management in modern buildings and industrial vehicles.
- المتانة: The covalent bonds formed by isocyanates create materials known for their extreme durability and resistance to structural fatigue, chemical corrosion, and mechanical wear.
- Flexibility and Stiffness: By adjusting the isocyanate formulation, engineers can create foams that are incredibly soft and flexible for seating, or panels that are extremely stiff to provide load-bearing structural integrity.
- Moisture Sensitivity: In their liquid state, isocyanates are highly moisture-sensitive, reacting with ambient humidity to release carbon dioxide gas. This property is actively harnessed to create the cellular structure of expanding foams.
How strong are isocyanate-derived products?
Isocyanate-derived composite materials exhibit immense strength and physical properties influenced by factors such as fiber form, fiber orientation, fiber content, and the specific chemical composition of the polymer matrix. Their behavior under physical strain, as seen in stress-strain curves, is absolutely crucial in determining their long-term strength and industrial durability. The chemical reinforcement equation, which involves particle modulus and covalent bond strength, strongly influences their overall structural integrity. Understanding the relationship between these chemical forces is essential in designing high-strength materials that resist impact and deformation.
How do isocyanates respond to heat?
The heat response of isocyanate-based materials varies greatly, largely influenced by their specific chemical constituents and the presence of flame-retardant additives. Advanced bio-composites and rigid insulating foams exhibit vastly improved thermal stability, maintaining their core properties even under high environmental temperatures. While standard polyurethanes can soften at extreme temperatures, highly cross-linked isocyanate resins exhibit unique thermal responses that resist melting, making them ideal for high-temperature automotive applications and electrical insulation.
Are isocyanate-derived materials resistant to corrosion?
A significant attribute of isocyanate-derived materials is their exceptional resistance to chemical and environmental corrosion, a characteristic that markedly influences their long-term physical properties. This resistance is due to the dense, highly cross-linked molecular structures, which provide an impenetrable protective barrier against water, salts, and industrial solvents. This rapid resistance to environmental degradation makes them ideal for applications requiring high strength and durability, such as marine coatings and underground pipeline insulation.
Recyclability and Sustainability of Isocyanates
Isocyanate-derived materials offer significant manufacturing advantages, being times stronger than traditional materials due to their dense matrix materials and highly tailored properties. They consist of individual polymer chains carefully aligned to enhance strength in specific directions, offering immense advantages in various long-lasting applications. However, handling the environmental impact of these powerful chemicals requires dedicated focus.
Can isocyanate-based materials be recycled?
Yes, but challenges in recycling these composite materials include their highly complex, thermosetting chemical form, which makes molecular separation incredibly difficult. Describing and understanding the properties of permanently cured fiber, including their locked orientation and structural content, significantly complicates the mechanical recycling challenge. Because the covalent bonds are extremely strong, traditional melting methods do not work. However, advanced chemical recycling processes called glycolysis and hydrolysis are used to break down isocyanate-derived foams back into their original polyol components, which can then be reused to create brand-new materials.
What challenges exist in recycling isocyanates?
The primary strength and modulus of the cured particles, along with complex chemical reinforcement equations, drastically affect material integrity during the recycling process. Considerations such as the original mold design, the presence of mixed materials (e.g., fabric bonded to foam), and potential longitudinal fractures during mechanical shredding pose significant technical challenges. Separating the pure polymer from metal inserts and chemical flame retardants requires expensive, energy-intensive processing facilities to ensure the recycled chemicals maintain their required industrial purity.
How sustainable are isocyanates?
Isocyanate materials offer strong sustainability metrics strictly due to their immense durability, lightweight nature, and high resource efficiency during their active lifespan. Because they create materials that are vastly stronger and lighter than traditional heavy metals, they significantly reduce fuel consumption when used in transportation. Furthermore, the exceptional insulating properties of isocyanate foams save millions of tons of greenhouse gas emissions by drastically reducing the energy required to heat and cool modern buildings. Sustainable modern design is guided by understanding these material properties to maximize energy savings over the product’s long lifespan.
What are the lifecycle assessment steps for isocyanates?
Lifecycle assessment (LCA) is a rigorous scientific process that completely evaluates the total environmental impact of isocyanate materials throughout their entire life cycle. It carefully considers factors such as the original chemical synthesis, matrix materials, and unique structural properties, as well as energy consumption during factory production. LCA evaluates the carbon footprint of the raw materials, the emissions generated during curing, and the long-term environmental effects of the finished product. Key aspects include understanding the material’s behavior under strain, energy savings during use, and the specific chemical phases required to safely recycle or dispose of the materials at the end of their functional life.
Innovations and Future Trends in Isocyanates
Future trends in isocyanate materials strongly emphasize improving chemical recycling and conducting in-depth lifecycle assessments to aggressively minimize overall environmental impact. Thoroughly understanding the material’s long journey from chemical production to final disposal is incredibly important for modern manufacturers.
What innovations are there in isocyanate technology?
Recent advancements in chemical technology have led to highly innovative recycling methods and comprehensive lifecycle tracking, significantly enhancing the global chemical industry’s sustainability. The development of advanced, non-toxic blocking agents, highly efficient carbon fiber recycling methods, and low-emission curing resins ranks among the most important recent innovations. These digital assessments accurately evaluate every stage of the materials’ lifecycle, rapidly enhancing understanding of environmental impact. New catalyst technologies also enable much faster, energy-efficient curing at lower factory temperatures.
What future trends impact isocyanate materials?
The rapid development of bio-based isocyanates represents a massive future trend currently transforming the industry. Researchers are successfully synthesizing isocyanate precursors from renewable plant-based oils rather than traditional petroleum sources. This significantly reduces the carbon footprint of the resulting polyurethanes. Additionally, the integration of advanced nanotechnology and smart responsive materials is impacting how these chemicals are utilized. By embedding conductive nanomaterials directly into the isocyanate matrix, the cured structures can actively monitor their own internal stress or change shape in response to thermal stimuli. Strategies for chemically repurposing these smart materials are being heavily explored to reduce industrial waste and enhance product durability in exciting new architectural and aerospace applications.
الخلاصة
Isocyanates are highly critical chemical components that continuously drive significant innovation across the modern industrial landscape. By masterfully reacting to form lightweight cellular cores and incredibly rigid outer skins, these engineered chemical compounds deliver unprecedented physical strength, environmental durability, and long-term cost-efficiency. Their diverse and demanding applications—ranging from critical aerospace weight reduction to advanced automotive safety and sustainable commercial construction—clearly demonstrate their remarkable chemical versatility and capacity to solve complex global engineering challenges. As we look toward the industrial future, ongoing technical advancements in bio-based chemistry, microcellular production, and closed-loop recycling methods will only further enhance the performance and sustainability of these vital materials. Ultimately, understanding and strategically leveraging the unique reactive properties of isocyanates will remain absolutely essential for engineers and manufacturers striving to build stronger, lighter, and vastly more energy-efficient products for generations to come.
TOPICS COVERED: Times stronger, matrix materials, material with properties, individual fibre, fiber direction, advantages of composites, natural composite, organic matrices, material properties, isotropic materials, orientation in material, affect properties of materials, orthotropic material, anisotropy of fiber composites, composites with metal fibers, carbon fibres, metal fibres, fibre reinforcement, fiber form, description of fiber, orientation of fibers, fibre content, fibre alignment, composite of cellulose fibres, strain curve, strain composites, strain of composite, modulus of particle, strength of particle, particle reinforcement equation, mould piece, upper mould, mould cavity, mould surface, longitudinal fracture, weak phase, strong phase, shear modulus, aramid fibres, natural fibres.